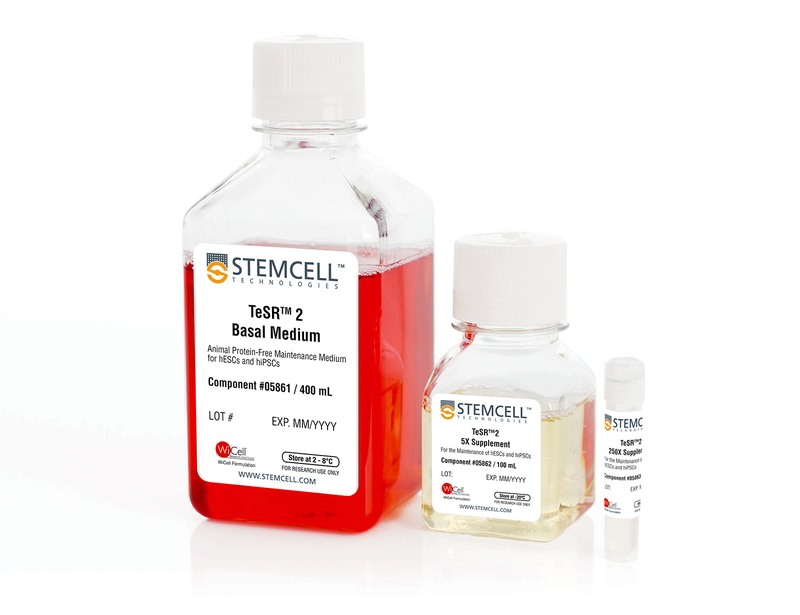
TeSR™2
Xeno-free, defined, feeder-free medium for maintenance of undifferentiated human ES and iPS cells
概要
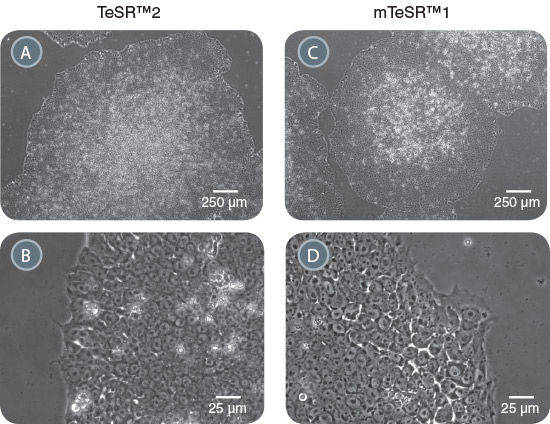
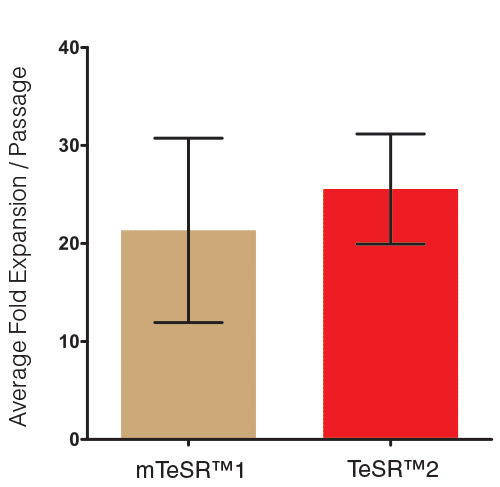
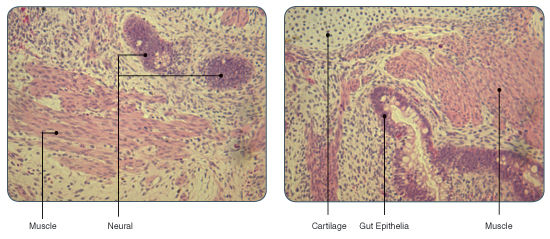
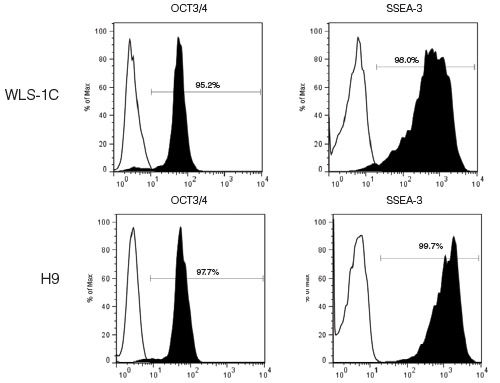
TeSR™2 is an improved version of mTeSR™1 which provides the same high-quality and robust system for feeder-free maintenance of human ES cells and iPS cells while enabling a more defined and xeno-free culture environment for basic research, stem cell banking, high-throughput studies and pre-clinical applications. Closely related to mTeSR™1, the most-published medium for the culture of human ES and iPS cells without feeders, TeSR™2 combines the advantages of a feeder-free culture system with the added value of being free of xenogenic components.
Cell Culture, Expansion, Maintenance
数据及文献
Publications (27)
Toxicology in vitro : an international journal published in association with BIBRA 2017 FEB
Development and characterization of a human embryonic stem cell-derived 3D neural tissue model for neurotoxicity testing.
Sandströ et al.
Abstract
Alternative models for more rapid compound safety testing are of increasing demand. With emerging techniques using human pluripotent stem cells, the possibility of generating human in vitro models has gained interest, as factors related to species differences could be potentially eliminated. When studying potential neurotoxic effects of a compound it is of crucial importance to have both neurons and glial cells. We have successfully developed a protocol for generating in vitro 3D human neural tissues, using neural progenitor cells derived from human embryonic stem cells. These 3D neural tissues can be maintained for two months and undergo progressive differentiation. We showed a gradual decreased expression of early neural lineage markers, paralleled by an increase in markers specific for mature neurons, astrocytes and oligodendrocytes. At the end of the two-month culture period the neural tissues not only displayed synapses and immature myelin sheaths around axons, but electrophysiological measurements also showed spontaneous activity. Neurotoxicity testing - comparing non-neurotoxic to known neurotoxic model compounds - showed an expected increase in the marker of astroglial reactivity after exposure to known neurotoxicants methylmercury and trimethyltin. Although further characterization and refinement of the model is required, these results indicate its potential usefulness for in vitro neurotoxicity testing.
Methods in molecular biology (Clifton, N.J.) 2016 APR
An Effective and Reliable Xeno-free Cryopreservation Protocol for Single Human Pluripotent Stem Cells.
Meng G et al.
Abstract
Efficient cryopreservation of human pluripotent stem cells (hPSCs) in chemically defined, xeno-free conditions is highly desirable for medical research and clinical applications such as cell-based therapies. Here we present a simple and effective slow freezing-rapid thawing protocol for the cryopreservation of feeder-free, single hPSCs. This cryopreservation protocol involves the supplementation of 10 % dimethyl sulfoxide (DMSO) and 10 $$M Rho-associated kinase inhibitor Y-27632 into two types of xeno-free, defined media supplements (Knockout Serum Replacement and TeSR2). High post-thaw cell recovery (˜90 %) and cell expansion (˜70 %) can be achieved using this protocol. The cryopreserved single cells retain the morphological characteristics of hPSCs and differentiation capabilities of pluripotent stem cells.
2016
Efficient Expansion of Dissociated Human Pluripotent Stem Cells Using a Synthetic Substrate.
Kawase E
Abstract
Human pluripotent stem cells (hPSCs), including human embryonic stem cells and human-induced pluripotent stem cells, are a renewable cell source for a wide range of applications in regenerative medicine and useful tools for human disease modeling and drug discovery. For these purposes, large numbers of high-quality cells are essential. Recently, we showed that a biological substrate, recombinant E8 fragments of laminin isoforms, sustains long-term self-renewal of hPSCs in defined, xeno-free medium with dissociated single-cell passaging. Here, we describe a modified culture system with similar performance to efficiently expand hPSCs under defined, xeno-free conditions using a non-biological synthetic substrate.
Methods in molecular biology (Clifton, N.J.) 2016
Efficient Production of Photoreceptor Precursor Cells from Human Embryonic Stem Cells.
Yanai A et al.
Abstract
Transplantation of photoreceptor precursor cells (PPCs) differentiated from human embryonic stem cells (hESCs) is a promising approach to treat common blinding diseases such as age-related macular degeneration and retinitis pigmentosa. However, existing PPC generation methods are inefficient. To enhance differentiation protocols for rapid and high-yield production of PPCs, we focused on optimizing the handling of the cells by including feeder-independent growth of hESCs, using size-controlled embryoid bodies (EBs), and addition of triiodothyronine (T3) and taurine to the differentiation medium, with subsequent removal of undifferentiated cells via negative cell-selection. Our novel protocol produces higher yields of PPCs than previously reported while reducing the time required for differentiation, which will help understand retinal diseases and facilitate large-scale preclinical trials.
2015
Microarray Approach to Identify the Signaling Network Responsible for Self-Renewal of Human Embryonic Stem Cells
Sato N and Brivanlou A
Abstract
Here we introduce the representative method to culture HESCs under the feeder and feeder-free conditions, the former of which is used to maintain or expand undifferentiated HESCs, and the latter can be used for the preparation of pure HESCs RNA samples, or for screening factors influential on self-renewal of HESCs. We also describe a protocol and tips for conducting gene chip analysis focusing on widely used Affymetrix Microarrays. These techniques will provide us unprecedented scale of biological information that would illuminate a key to decipher complex signaling networks controlling pluripotency.
Methods in molecular biology (Clifton, N.J.) 2015
Generation, Expansion, and Differentiation of Human Pluripotent Stem Cell (hPSC) Derived Neural Progenitor Cells (NPCs).
Brafman DA
Abstract
Human pluripotent stem cell (hPSC)-derived neural progenitor cells (NPCs), a multipotent cell population that is capable of near indefinite expansion and subsequent differentiation into the various cell types that comprise the central nervous system (CNS), could provide an unlimited source of cells for neural-related cell-based therapies and disease modeling. However, the use of NPCs for the study and treatment of a variety of debilitating neurological diseases requires the development of scalable and reproducible protocols for their generation, expansion, characterization, and neuronal differentiation. Here, we describe a serum-free method for the stepwise generation of NPCs from hPSCs through the sequential formation of embryoid bodies (EBs) and neuro-epithelial-like rosettes. NPCs isolated from neural rosette cultures can be homogenously expanded while maintaining high expression of pan-neural markers such as SOX1, SOX2, and Nestin. Finally, this protocol allows for the robust differentiation of NPCs into microtubule-associated protein 2 (MAP2) and β-Tubulin-III (β3T) positive neurons.
View All Publications